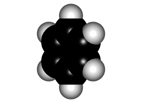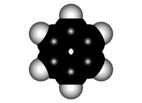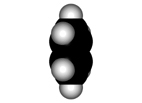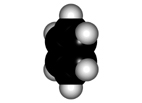
Faraday isolated benzene in 1825 for the first time from gas lighting and obtained "CH" as its empirical formula.
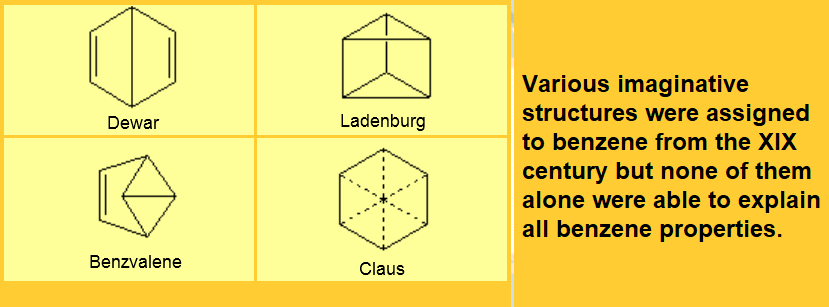
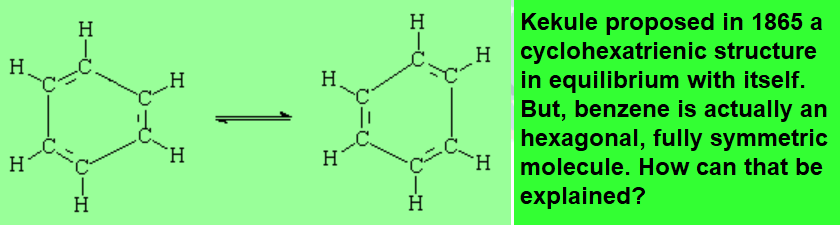
In 1834 Mitscherlich determined its molecular formula as C6H6, that was difficult to explain at that time because it seemed to violate carbon tetrevalency.
Michael Faraday FRS (22 September 1791 – 25 August 1867) was an English scientist who contributed to the study of electromagnetism and electrochemistry. His main discoveries include the principles underlying electromagnetic induction, diamagnetism and electrolysis.
Eilhard Mitscherlich (7 January 1794 – 28 August 1863) was a German chemist, who is perhaps best remembered today for his discovery of the phenomenon of isomorphism (crystallography) in 1819.
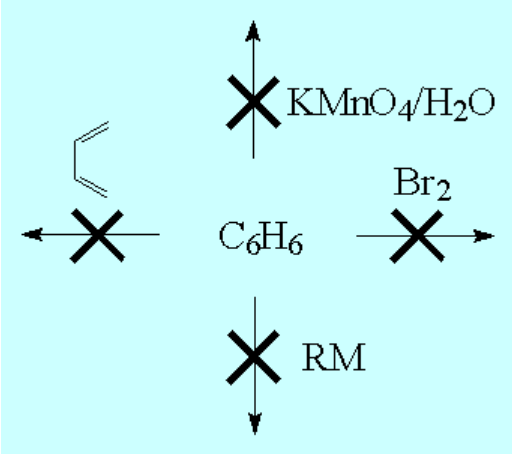
Benzene is a highly unsaturated hydrocarbon, very special and very stable, that does not undergo the usual reactions of alkenes or alkynes.
That's puzzling!!!
Why is that?
The heat released when benzene is hydrogenated is much lower than that anticipated if the three double C=C bonds behaved as "normal" alkenes.
This experimental fact indicates that benzene is much more stable than expected.